New Real-Time Imaging, Non-Invasive Cell Labeling Technology Discovered
2 Cell stage embryos have only one of these cells
It will evolve into the 80% embryonic host, the future ectoderm, the endoderm (yolk sac) and the trophoblast (placenta).
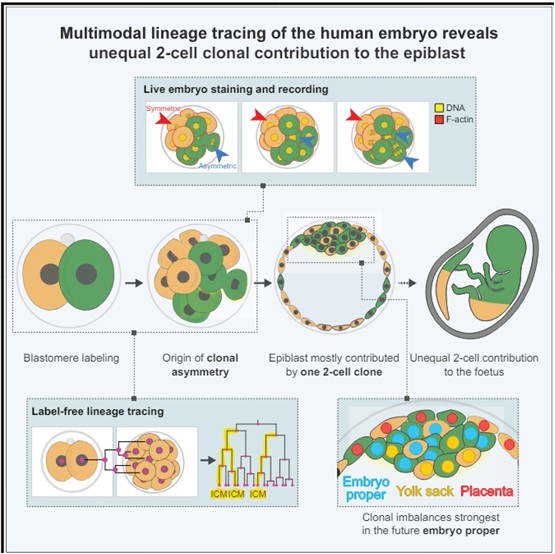
Labeling and live imaging of human embryos reveal that the majority of the future body originates, mostly, from one of the 2-cell stage blastomeres. Descendants of the first 2-cell stage blastomere to divide contribute more asymmetric divisions at the 8-cell stage, which generate the small number of founding epiblast cells before implantation. founding epiblast cells before implantation.
The first two blastomeres contribute unequally to the human embryo
SUMMARY
Retrospective lineage reconstruction of humans predicts that dramatic clonal imbalances in the body can be traced to the 2-cell stage embryo. However, whether and how such clonal asymmetries arise in the embryo is unclear. Here, we performed prospective lineage tracing of human embryos using live imaging, non-invasive cell labeling, and computational predictive imaging. imaging, non-invasive cell labeling, and computational predictions to determine the contribution of each 2-cell stage blastomere to the epiblast ( body), hypoblast (yolk sac), and trophectoderm (placenta). We show that the majority of epiblast cells originate from only one blastomere of the 2-cell stage embryo. We observe that only one to three cells become internalized at the 8-to-16-cell stage. Moreover, these internalized cells are more frequently derived from the first cell to divide at the 2-cell stage. -We propose that cell division dynamics and a cell internalization bottleneck in the early embryo establish asymmetry in the clonal composition of the future human body. composition of the future human body.